Vol 66, No 1 (2021)
- Year: 2021
- Published: 07.03.2021
- Articles: 9
- URL: https://virusjour.crie.ru/jour/issue/view/48
Full Issue
REVIEWS
Ebola virus (Filoviridae: Ebolavirus: Zaire ebolavirus): fatal adaptation mutations
Abstract
Ebola virus disease (EVD) (former Ebola hemorrhagic fever) is one of the most dangerous infectious diseases affecting humans and primates. Since the identification of the first outbreak in 1976, there have been more than 25 outbreaks worldwide, the largest of which escalated into an epidemic in 2014-2016 and caused the death of more than 11,000 people. There are currently 2 independent outbreaks of this disease in the eastern and western parts of the Democratic Republic of the Congo (DRC) at the same time. Bats (Microchiroptera) are supposed to be the natural reservoir of EVD, but the infectious agent has not yet been isolated from them. Most animal viruses are unable to replicate in humans. They have to develop adaptive mutations (AM) to become infectious for humans. In this review based on the results of a number of studies, we hypothesize that the formation of AM occurs directly in the human and primate population and subsequently leads to the development of EVD outbreaks.
 7-16
7-16


Rapid diagnostics of novel coronavirus infection by loop-mediated isothermal amplification
Abstract
This review presents the basic principles of application of the loop-mediated isothermal amplification (LAMP) reaction for the rapid diagnosis of coronavirus infection caused by SARS-CoV-2. The basic technical details of the method, and the most popular approaches of specific and non-specific detection of amplification products are briefly described. We also discuss the first published works on the use of the method for the detection of the nucleic acid of the SARS-CoV-2 virus, including those being developed in the Russian Federation. For commercially available and published LAMP-based assays, the main analytical characteristics of the tests are listed, which are often comparable to those based on the method of reverse transcription polymerase chain reaction (RT-PCR), and in some cases are even superior. The advantages and limitations of this promising methodology in comparison to other methods of molecular diagnostics, primarily RT-PCR, are discussed, as well as the prospects for the development of technology for the detection of other infectious agents.
 17-28
17-28


ORIGINAL RESEARCHES
Biological characteristics and permissiveness to viruses of diploid kidney cells strain from the bat Nathusius’ pipistrelle (Pipistrellus nathusii Keyserling & Blasius, 1839; Chiroptera: Microchiroptera: Vespertilionidae)
Abstract
Introduction. Bats are an epidemiologically important natural reservoir of viruses of various taxonomic groups, including causative agents of especially dangerous infections of humans and animals. Considering the relevance of arbovirus infections, it seems advisable to study the spectrum of the sensitivity of cells derived from bats inhabiting and migrating on the territory of the Russian Federation to causative agents of vector-borne diseases of animals.
The study aimed to obtain a diploid strain of cells from renal tissue of bats Pipistrellus nathusii and to investigate its biological characteristics, as well as to assess its permissiveness for bluetongue (BTV); Rift Valley fever (RVFV); lumpy skin disease (LSDV); rabbit myxoma (Myxomatosis cuniculi); rabbit, or Shope fibroma (RFV); African horse sickness (AHSV) and African swine fever (ASFV) viruses.
Material and methods. There were 2 clinically healthy male individuals of P. nathusii who taken as donors of organs. To obtain diploid kidney cell culture strain and to study its properties, the level of the 6th passage was investigated by conventional cytological, virological, and molecular methods. The permissiveness of the obtained cell culture for BTV, RVFV, LSDV, Myxomatosis cuniculi, RFV, AHSV and ASFV was determined.
Results. The formation of a confluent monolayer was observed after 72 hours, while the proliferation index was 2.7-3.3. The cell monolayer had been maintained without changing the medium for 45 days (observation period). The stability of the karyotype had been demonstrated in continuous subculturing at the 36th passage. The cell culture named «Diploid cell line Pipistrellus nathusii kidney», and its permissiveness to BTV, RVFV, LSDV and Myxomatosis cuniculi had been demonstrated.
Discussion. The sensitivity of the strain to BTV and RVFV is consistent with the data on the identification of reovirus and RVFV in Egyptian fruit bats (Rousettus aegyptiacus), and its permissiveness for LSDV and rabbits myxoma virus is consistent with the results of detection of poxviruses in big brown bat (Eptesicus fuscus). Conclusion. A diploid kidney cell strain derived from P. nathusii was obtained and certified. Its permissiveness to BTV, RVFV, LSDV and rabbits myxoma viruses makes it possible to use this strain for isolation and studies of these viruses. Reproduction of the viruses in diploid kidney cells strain derived from P. nathusii living and migrating in the European part of the Russian Federation indicates their potential role in the epidemiology of significant infections, especially transmissible ones.
 29-39
29-39


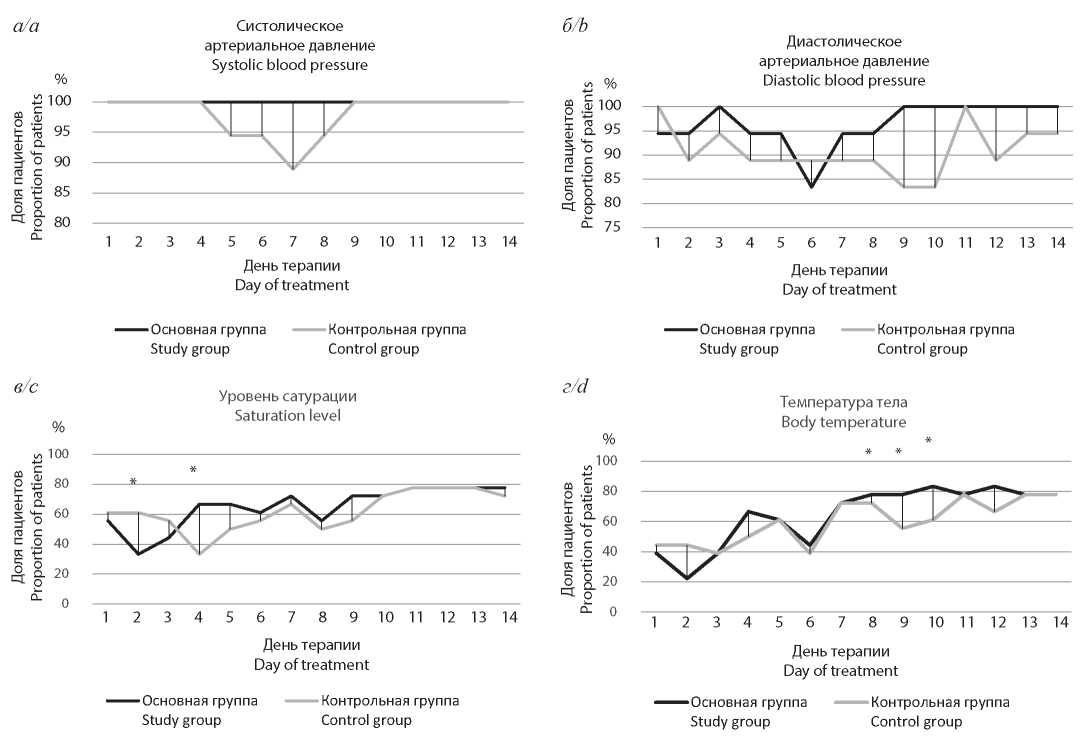
Effect of anticoagulant therapy on the course of COVID-19 in comorbid patients
Abstract
Introduction. Analysis of the pathogenesis of coronavirus infection caused SARS-CoV-2 indicates a significant impact of hemorheological disorders on its course and outcomes. It is known that chronic cardiovascular diseases are associated with the risk of severe course and lethal outcomes both in COVID-19 and other infectious diseases. Therefore, in each case it is necessary to study the interaction and mutual influence of different components of the treatment program prescribed to such patients.
The purpose of this work was to evaluate the effect of coagulation activity on the course of a novel coronavirus infection (COVID-19) and to justify the management of comorbid patients having been received novel oral anticoagulants (NOACs) in previously selected doses according to indications in concomitant somatic diseases.
Material and methods. Total 76 cases of confirmed coronavirus infection in patients who had been received initial therapy on an outpatient basis were analyzed. 26 patients who received NOACs (rivaroxaban, apixaban, dabigatran) made up the main group and 50 - the comparison (control) group in which patients had not been administered any drugs that affect blood clotting until the episode of COVID-19. All patients have been prescribed therapy following the Provisional guidelines «Prevention, diagnosis and treatment of coronavirus infection (COVID-19)» (https://static-0.minzdrav.gov.ru/system/attachments/attaches/).
Results and discussion. The number of hospitalizations was significantly fewer in the group of patients who had been received NOACs (19 vs. 66% in the control group). No deaths or cases of severe respiratory and/or renal failure were observed in the main group, while adverse outcomes were noted in 14% of patients who had not been administered these drugs.
Conclusion. Taking NOACs reduces the probability of severe course and adverse outcomes in the development of coronavirus infection caused by SARS-CoV-2, which indicates a significant contribution of coagulation mechanisms to the pathogenesis in COVID-19. There were no indications for drug replacement and correction of anticoagulant therapy regimens in patients who received adequate therapy with oral anticoagulants for treating a non-severe form of coronavirus infection in ambulatory patient settings.
 40-46
40-46


Interferon gamma in the treatment of patients with moderate COVID-19
Abstract
Introduction. Interferons are produced in response to the presence of pathogens in cells and are responsible for the proper formation of immune reaction. Preliminary data obtained in studies of properties of recombinant interferon gamma (IFN-γ) that involved patients with community-acquired pneumonia (including bacterial), acute respiratory viral infection (ARVI), influenza and new coronavirus infection have shown promising results.
The purpose of the study was to assess the effect of subcutaneous administration of IFN-γ in patients with viral pneumonia on the changes of vital signs and the duration of hospital stay.
Material and methods. An open-label, randomized, low-interventional study included patients with moderate new coronavirus infection COVID-19 over 18 years of age of both sexes. IFN-γ 500,000 IU was administered s/c, daily, once a day, during 5 days.
Results. IFN-y in addition to complex therapy of the disease resulted in more favorable changes in the stabilization of vital signs, as well as in reduced length of fever and hospital stay by 2 days what allows suggesting a positive effect of this substance on the recovery processes in patients with moderate COVID-19. Special emphasis should be made to the fact that patients who received recombinant IFN- γ experienced no progression of respiratory failure and required no transfer to intensive care unit.
Discussion. This study confirms earlier obtained data on the positive effect of IFN-y on the rate of clinical stabilization and recovery of patients with community-acquired pneumonia and viral infections. Presented results are limited to a small number of patients; further study of drug properties in post-marketing studies is required.
Conclusion. Progress in the treatment of patients with moderate COVID-19 by adding recombinant IFN-γ to the complex therapy may reasonably expand the range of existing treatment options for this infection.
 47-54
47-54


Synthesis and characterization of human rotavirus A (Reoviridae: Sedoreovirinae: Rotavirus: Rotavirus A) virus-like particles
Abstract
Introduction. Rotavirus infection is the leading cause of acute gastroenteritis among infants. The development of new vaccines against rotavirus A is urgent because the virus has many genotypes, some of which have regional prevalence. Virus-like particles (VLP) is a promising way to create effective and safe vaccine preparations.
The purpose of the study is to develop the technology for the production of VLP, containing VP2, VP4, VP6 and VP7 of viral genotypes prevalent on the territory of the Russian Federation, and to give its molecular genetic and virological characteristics.
Material and methods. The virulent strain Wa G1P[8] of human RV A adapted to MARC-145 cell culture has been used. It was cultured and purified according to the method described by the authors earlier. Standard molecular genetic and cytological methods were used: gene synthesis; cloning into transfer plasmids; recombinant baculoviruses production in Bac-to-Bac expression system; VLP production in the insect cells; centrifugation in sucrose solution; enzyme-linked immunosorbent assay (ELISA); electron microscopy (EM); polyacrylamide gel electrophoresis (SDS-PAGE) and western blot analysis.
Results. VP4 and VP7 of the six most represented in Russia genotypes: G1, G2, G4, G9, P4, P8, as well as VP2 and VP6 were selected for VLP production. Recombinant baculoviruses were obtained with codon frequencies optimized for insect cells. Cabbage loopper (Trichoplusia ni) cell culture was coinfected with different combinations of baculoviruses, and VLP consisting of 2-4 proteins were produced. VLP were purified by centrifugation. The size and morphology of the particles matched the rotavirus A virion (by EM). The presence of rotavirus A proteins in VLP was confirmed by the ELISA, SDS-PAGE and western blot analysis.
Conclusion. The technology for the synthesis of three-layer VLP consisting of VP2, VP4, VP6 and VP7 has been developed and optimized. The resulting VLP composition represents 6 serotypes of VP4 and VP7, which are most represented on the territory of Russia, and can be used for vaccine development.
 55-64
55-64


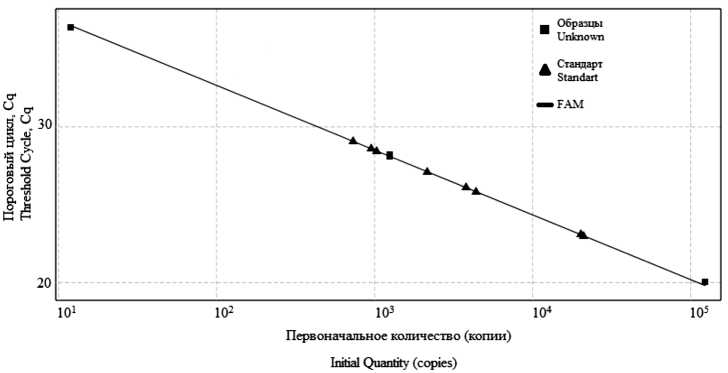
Real-time PCR assay development for the control of vaccine against hemorrhagic fever with renal syndrome
Abstract
Introduction. Hemorrhagic fever with renal syndrome (HFRS) holds a leading place among natural focal human diseases in Russian Federation. There is no etiotropic therapy for the disease now. The vaccine prophylaxis is the most effective method to control this infection. The main criteria for inactivated vaccines evaluation are its immunogenicity and specific activity.
The study purposes were to develop a sensitive and specific real-time PCR method for viral RNA quantification in the inactivated vaccine and to study the correlation between the viral RNA amount and vaccine immunogenicity.
Material and methods. L-segment fragments of the Puumala, Hantaan, and Sochi vaccine strains were selected as diagnostic targets for oligonucleotides and fluorescent probes. The immunogenicity of experimental vaccines was determined by the induction of neutralizing antibodies in BALB/c mice.
Results. A highly specific, sensitive and reproducible real-time PCR method has been developed. The analytical sensitivity was 1.24 ± 1.5 x 102 copies/ml for Puumala virus; 1.16 ± 1.4 * 102 copies/ml for Hantaan; 1.32 ± 1.8 * 102 copies/ ml for Sochi, with a virus content of 1.5 ± 0.5 lg FFU/ml; 1.8 ± 0.5 lg FFU/ml and 2.2 ± 0.5 lg FFU/ml, respectively. The viral RNA amount in experimental vaccine preparations inactivated with β-propiolactone was proportional to the neutralizing antibodies titer observed in mice following the immunization.
Discussion. It was found that different virus inactivators differently affects the detected viral RNA amount, but not the vaccine immunogenicity, which indicates the same degree of the immunogenic proteins damage. The direct relationship between the viral RNA copy number and vaccine immunogenicity makes it possible to use this criterion for vaccine dosage preparation.
Conclusion. The developed method for viral RNA quantification is a promising tool for the specific activity control of the HFRS vaccine.
 65-73
65-73


Clinical course and outcomes of chronic viral hepatitis D in patients from Republic of Tuva as endemic region
Abstract
Introduction. Hepatitis D (delta, 5) is caused by an RNA virus (hepatitis D virus, HDV) from genus Deltavirus, and is the most severe and difficult to treat disease among both viral hepatitis and infectious diseases in general. The development of HDV infection in the host organism is possible only in the presence of hepatitis B virus (HBV). Coinfection with HBV and HDV is associated with a more rapid progression of chronic viral hepatitis (CVH) to liver cirrhosis (LC) and an unfavorable outcome in comparison with HBV monoinfection. Data on the influence of clinical, biochemical and virological factors on the infectious process in patients with hepatitis D are limited due to the insufficient amount of research on this theme.
The study aimed to determine demographic, clinical, biochemical, and virological factors influencing the course and progression of CVH D in patients followed during 10 years, residing in the territory of the Tuva Republic, one of the endemic regions of the Russian Federation.
Material and methods. Changes in clinical and laboratory parameters were analyzed in dynamics in 121 HDV infected patients with a different course of the disease, who were under observation from 2009 to 2019. Three groups of patients were identified: group 1 - 61 patients with disease progression of chronic hepatitis to LC (Child-Pugh class B-C), group 2 - 49 patients with non-progressive chronic hepatitis, and group 3 - 11 patients with slowly progressive LC (class A). Demographic data, the presence of detectable HBV DNA, indicators of the functional state of the liver: alanine aminotransferase (ALT/GPT), aspartate aminotransferase (AST/GOT), alkaline phosphatase (ALP), gamma-glutamyltransferase (GGT), and total bilirubin content were analyzed. The severity of hepatic encephalopathy was assessed by the duration of the numbers connection test (NCT).
Results. All patients belonged to the same ethnic group (Tuvinians), were infected with HDV genotype 1 and were positive for HDV RNA throughout the entire follow-up period. There were no significant differences in sex ratio and mean age at the time of inclusion in the study between the groups. In group 1, the average number of years from inclusion in the study to the formation of LC was 3.65 ± 2.3 years, years to the lethal outcome: 4.5 ± 3 years. Significantly higher levels of AST/GOT, ALP, GGT, total bilirubin (TB) and NCT grade were found in group 1 compared to group 2. ALT/GPT levels did not differ significantly in these groups. When comparing groups with disease progression and slowly progressive LC (groups 1 and 3), no significant differences were found in any of the clinical and biochemical parameters. ALT/GPT, GGT, TB and NCT values were significantly higher in patients with slowly progressive LC (group 3) compared to group 2. No differences in AST/GOT and ALP levels were found between these groups. Detectable HBV DNA was significantly more frequent in patients with progressive disease and with chronic viral hepatitis than in patients with slowly progressive LC. There were no significant differences in the frequency of HBV DNA detection in patients from groups 1 and 2.
Conclusion. The results obtained on a relatively homogeneous cohort demonstrated that age and gender are not the factors influencing the progression of chronic viral hepatitis D to cirrhosis. The lack of detectable HBV DNA is associated with the slow progression of LC. The revealed differences in clinical and biochemical parameters reflect the degree of functional liver damage in chronic viral hepatitis D and HDV-associated cirrhosis.
 74-83
74-83


BOOK REVIEW
 84
84